LDN Community
Join our community here.
Dickson Chemist developed a new formulation of LDN which will hopefully improve the outcomes for patients. This new formulation is a more concentrated liquid in the form of drops which are administered under the tongue and absorbed directly from the mucosal lining of the mouth. This sublingual route produces a more rapid absorption bypassing the liver and digestive tract. There are several potential advantages to this, the drug may be more effective and will not be affected by the presence of food stuffs or the hostile environment of the gut. Each drop contains 0.5 mg of LDN so the dose is easy to calculate and avoids the use of a syringe to draw up the dose which can be problematic. The bottle is not being repeatedly opened and closed so there is less of a chance that the liquid will oxidise from contact with the air which can cause it to become bitter and “go off”. Also there is much reduced chance of infection being inadvertently introduced into the mixture from contact with the syringe.
One of the potential advantages of sublingual drops is in patients whose dose is limited by nausea. One such patient was the first to be tried on sublingual LDN, a 33 year old male with severe fibromyalgia. This patient was forced to give up work due to the severity of his pain and walks with the aid of a walking stick. His symptoms have been very difficult to treat and it has been a source of frustration to both doctor and patient that we could not maximise his LDN dose due to nausea. His dose of LDN had reached 10mg twice daily and still we were not improving his pain or fatigue. It was suggested that he try this new formulation of LDN and he readily agreed.
After one month on the same dose he reported that his nausea was virtually gone and surprisingly that his pain and fatigue was improved. This was a pleasant surprise as my aim was primarily to reduce his nausea to enable further dose increases in an effort to improve his symptoms. The symptomatic improvement suggested that this route of administration was producing better absorption with a greater bioavailability. Effectively more drug was reaching the blood stream when absorbed from under the tongue rather than from the stomach. Drugs absorbed from the digestive system are subsequently metabolised in the liver in what is known as “first pass metabolism” which can alter and degrade the drug. Absorption from the mucous membranes of the mouth avoids this type of metabolism and so more drug will get directly into the blood stream. Drugs in the stomach can also be degraded by the stomach acid, bile and several enzymes so the sublingual route is more likely to deliver the pure drug to the system.
The picture below shows the difference between a swallowed oral dose and a sublingual dose for another drug which mirrors the situation with this new formulation of LDN.
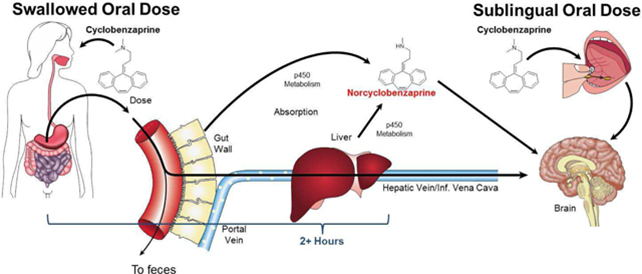
Regardless of the theory, this patient was delighted with the effect which he described as “one hundred times better”. We have been able to increase his dose to 11mg twice daily and can see some light at the end of a very dark tunnel. Beyond this case this new form of the drug means we could be at the beginning of another exciting new chapter for LDN.







